Insightsintothecellularoriginofcervicalcancer
ResearchteamledbyCAUSeniorProfessorMeyerdiscoverscentralmechanismwhichinfluencesthestartofthepathogenesisofcancer.
Cervicalcanceristhefourthmostcommontypeofcancerinwomen.Morethan90%ofcasesdevelopinthetransitionzonebetweentheinnercervix(endocervix)andoutercervix(ectocervix)acriticalareainwhichthereisanincreasedincidenceofinfectionsandinflammation.Cancerisoftenprecededbymetaplasiabeforeitdevelopsinthetransitionzone.Thisisaprocessinwhichthecelltypesthatarenormallypresentinatissuearereplacedbyothercelltypes.Thetissuetherebyadaptstochangesinthelocalenvironment,whichcouldbecausedbydietorexternalfactorssuchasinfections.Despitetheirenormousimportanceforthepathogenesisofcancer,itwaspreviouslyunclearexactlyhowmetaplasiasarise.ProfessorThomasF.Meyer,seniorprofessoratKielUniversity(CAU)andamemberoftheClusterofExcellence"PrecisionMedicineinChronicInflammation"(PMI),togetherwithhisformerresearchteamfromtheMaxPlanckInstituteforInfectionBiology(MPIIB)inBerlinandincooperationwithadditionalresearchers,hasnowpresentedimportantnewinsightsintotheirorigin.TheteamrecentlypublishedtheirresultsintherenownedscientificjournalNatureCellBiology.
Thecelltypesoftheneighboringareasmeetinthetransitionzone
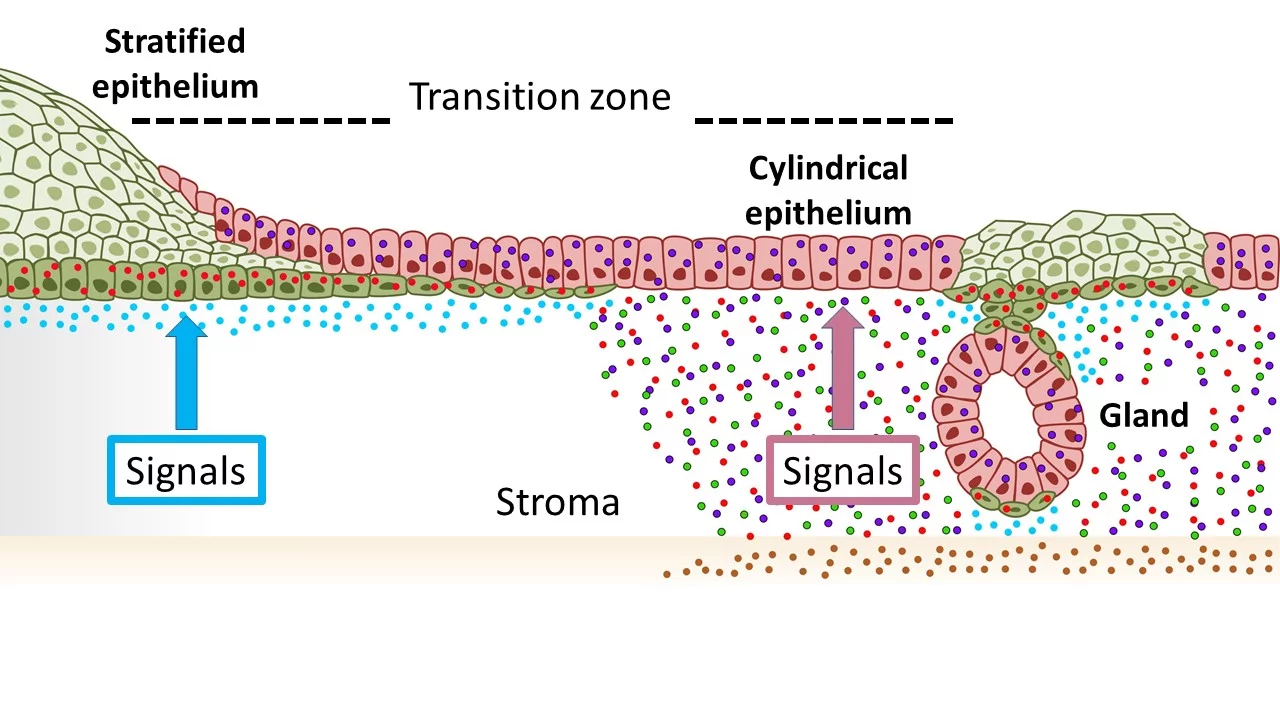
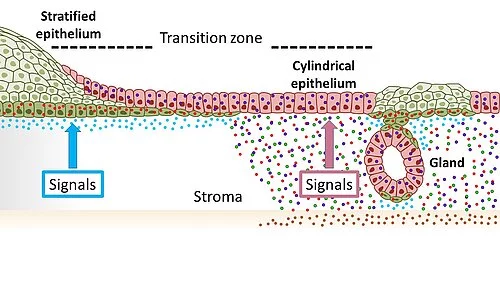
"Wefirstanalyzedthetransitionzoneinthecervixfromtheinnertotheoutercervixindetailatthecellularlevel.Previously,itwasscientificallycontroversialwhichcelltypesmakeupthistransitionzone,"saidthecoordinatingauthorandrecentlyappointedCAUSeniorProfessorMeyer,whocompletedthemajorityoftheresearchattheMaxPlanckInstituteforInfectionBiologyinBerlin.Todoso,theyessentiallyreconstructedthetissueinthelaboratoryusingstemcellsfromtheoriginaltissue,intheformofso-calledorganoids."Wewereabletoshowthatthetransitionzonedoesnotconsistofitsowncelltype-aswaswidelyassumed-butratherthatthetwocelltypesoftheneighboringareasmeethere,"saidMeyer.Morespecifically,itistheso-calledsquamousepithelialcellswhichcovertheoutercervix,andtheso-calledcolumnarepithelialcellswhicharefoundintheinnercervix. 
Opposingsignalsregulatewhichcellisactivewhere
"Inordertounderstandhowthetwocelltypesleadtometaplasiasandultimatelytocancercells,itisimportanttofindouthowthesecellsandtheirrespectivedevelopmentsareregulated,"explainedthefirstauthorofthepublication,Dr.CindrillaChumduri,formerresearchassociateattheMPIIBandrecentlyappointedgroupleaderattheBiocenteroftheUniversityofWrzburg."Herewemadeasurprisingdiscovery.Theneighboringepithelialcelltypes,whichmeetinthetransitionzone,aredependentonopposingcellularbiologicalsignals,"continuedChumduri.Accordingly,thecolumnarepithelialcellsoftheinnercervixrequireaspecificsignaltosurvive,whichissentviathebiologicallyimportantso-calledWntsignalingpathway,whereasthesquamousepithelialcellsoftheoutercervixaresuppressedbypreciselythissignal.
Shiftingofthesignalsleadstometaplasia
"Wewereabletoshowthatagradient,i.e.differentlevelsofoccurrenceofthesesignalsinthecervixintheimmediateenvironmentofthecells,ensuresthattherespectiveneighboringbutdifferentcelltypesaremaintained.Ifthesesignalsshiftinonedirection,thenonetypeoftissuealsoshiftsinthedirectionoftheothersometaplasiaoccurs,"summarizedfirstauthorDr.RajendraGurumurthy,alsoaformeremployeeattheMPIIB.TheresearcherswereabletoshowinamousemodelthatavitaminAdeficiency,forexample,leadstosuchasignalshift.Asaresult,squamousepithelialcellswereactivatedintheinnercervix,andreplacedthecolumnarepithelialcellswhicharenormallypresentinthisarea.
"Ourfindingshelptounderstandthemechanismsofthepathogenesisofcancerinthesetransitionzonesmoreprecisely.Theycanalsohelptoimprovetheearlydetectionofcervicalcancer,andtodevelopprecisestrategiesforpreventionandimprovedtreatments,"saidMeyer."Asthenextstep,wearenowtryingtobetterunderstandtheexternalinfluencesandprocesseswhichleadtoasignalchangeintheimmediatecellenvironment,andthustometaplasiaformation.Preliminaryfindingsalsosuggestapossibleroleofinflammationhere.We'llinvestigatethisinfuturestudiesinKiel,alsotogetherwithourcolleaguesattheClusterofExcellencePMI,"addedMeyer.
Scientificcontact:
Prof.Dr.ThomasF.Meyer
InfectionOncologyworkinggroup
InstituteofClinicalMolecularBiology(IKMB)
KielUniversityandtheUniversityMedicalCenterSchleswig-Holstein
t.meyer@ikmb.uni-kiel.de
Illustrationofthetissueinthecervixwithsquamousepithelia(green)oftheoutercervix(ectocervical)andcolumnarepithelia(red)oftheinnercervix(endocervical)mergingatthetransitionzone.TheopposingWntsignalingenvironmentmaintainsthisdistribution.AchangetowardsignalsinwhichWntsignalingissuppressedintheendocervicalstromapromotesthedevelopmentofsquamousmetaplasia.
Originalpublication:
C.Chumduri*,R.K.Gurumurthy*T.F.Meyer:OpposingWntsignalsregulatecervicalsquamocolumnarhomeostasisandemergenceofmetaplasia.NatureCellBiology(2021).https://doi.org/10.1038/s41556-020-00619-0
*Theseauthorscontributedequallytothework.
AbouttheClusterofExcellencePMI
TheClusterofExcellence"PrecisionMedicineinChronicInflammation"(PMI)isbeingfundedfrom2019to2025throughtheGermanExcellenceStrategy(ExStra).Itsucceedsthe"InflammationatInterfacesCluster,whichwasalreadyfundedintwoperiodsoftheExcellenceInitiative(2007-2018).Around300membersfromeightinstitutionsatfourlocationsareinvolved:Kiel(KielUniversity,UniversityMedicalCenterSchleswig-Holstein(UKSH),MuthesiusUniversityofFineArtsandDesign,KielInstitutefortheWorldEconomy(IfW),LeibnizInstituteforScienceandMathematicsEducation(IPN)),Lbeck(UniversityofLbeck,UniversityMedicalCenterSchleswig-Holstein(UKSH)),Pln(MaxPlanckInstituteforEvolutionaryBiology)andBorstel(ResearchCenterBorstel-LeibnizLungCenter).
Thegoalistotranslateinterdisciplinaryresearchfindingsonchronicinflammatorydiseasesofbarrierorganstohealthcaremoreintensively,aswellastofulfilpreviouslyunsatisfiedneedsofthepatients.Threepointsareimportantinthecontextofsuccessfultreatment,andarethereforeattheheartofPMIresearch:theearlydetectionofchronicinflammatorydiseases,thepredictionofdiseaseprogressionandcomplications,andthepredictionofindividualresponsestotreatment.
Pressoffice
fbuhse@uv.uni-kiel.de+49(0)431/8804682https://precisionmedicine.de
ClusterofExcellence "PrecisionMedicineinChronicInflammation"
ScientificOffice
Head:Dr.habil.SusanneHolsteinPostal
Christian-Albrechts-Platz4,24118Kiel,Germany
Contact:SonjaPetermann
 +49(0)431880-4850,fax:+49(0)431880-4894
spetermann@uv.uni-kiel.de
Twitter:PMI@medinflame