Insights into the cellular origin of cervical cancer
Research team led by CAU Senior Professor Meyer discovers central mechanism which influences the start of the pathogenesis of cancer.
Cervical cancer is the fourth most common type of cancer in women. More than 90% of cases develop in the transition zone between the inner cervix (endocervix) and outer cervix (ectocervix) – a critical area in which there is an increased incidence of infections and inflammation. Cancer is often preceded by metaplasia before it develops in the transition zone. This is a process in which the cell types that are normally present in a tissue are replaced by other cell types. The tissue thereby adapts to changes in the local environment, which could be caused by diet or external factors such as infections. Despite their enormous importance for the pathogenesis of cancer, it was previously unclear exactly how metaplasias arise. Professor Thomas F. Meyer, senior professor at Kiel University (CAU) and a member of the Cluster of Excellence "Precision Medicine in Chronic Inflammation" (PMI), together with his former research team from the Max Planck Institute for Infection Biology (MPIIB) in Berlin and in cooperation with additional researchers, has now presented important new insights into their origin. The team recently published their results in the renowned scientific journal Nature Cell Biology.
The cell types of the neighboring areas meet in the transition zone
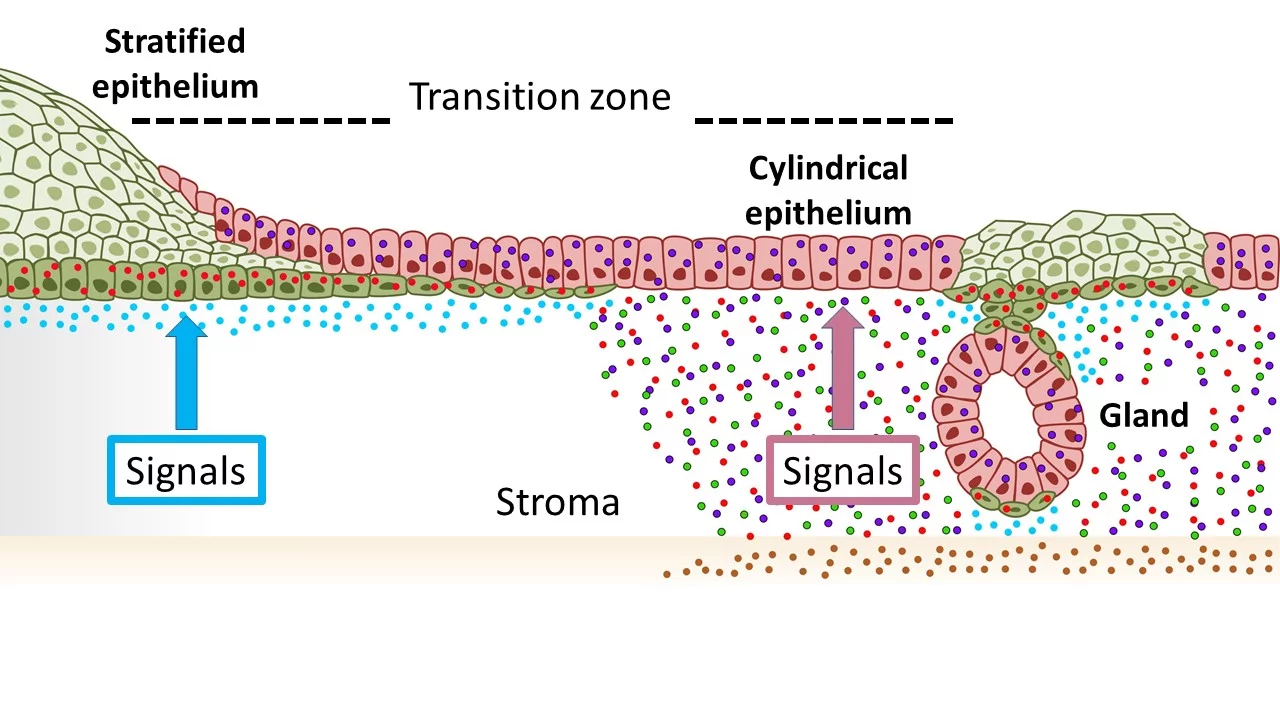
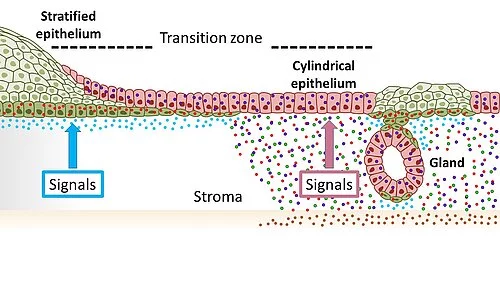
"We first analyzed the transition zone in the cervix from the inner to the outer cervix in detail at the cellular level. Previously, it was scientifically controversial which cell types make up this transition zone," said the coordinating author and recently appointed CAU Senior Professor Meyer, who completed the majority of the research at the Max Planck Institute for Infection Biology in Berlin. To do so, they essentially reconstructed the tissue in the laboratory using stem cells from the original tissue, in the form of so-called organoids. "We were able to show that the transition zone does not consist of its own cell type - as was widely assumed - but rather that the two cell types of the neighboring areas meet here," said Meyer. More specifically, it is the so-called squamous epithelial cells which cover the outer cervix, and the so-called columnar epithelial cells which are found in the inner cervix.
Opposing signals regulate which cell is active where
"In order to understand how the two cell types lead to metaplasias and ultimately to cancer cells, it is important to find out how these cells and their respective developments are regulated," explained the first author of the publication, Dr. Cindrilla Chumduri, former research associate at the MPIIB and recently appointed group leader at the Biocenter of the University of Würzburg. "Here we made a surprising discovery. The neighboring epithelial cell types, which meet in the transition zone, are dependent on opposing cellular biological signals," continued Chumduri. Accordingly, the columnar epithelial cells of the inner cervix require a specific signal to survive, which is sent via the biologically important so-called Wnt signaling pathway, whereas the squamous epithelial cells of the outer cervix are suppressed by precisely this signal.
Shifting of the signals leads to metaplasia
"We were able to show that a gradient, i.e. different levels of occurrence of these signals in the cervix in the immediate environment of the cells, ensures that the respective neighboring but different cell types are maintained. If these signals shift in one direction, then one type of tissue also shifts in the direction of the other – so metaplasia occurs," summarized first author Dr. Rajendra Gurumurthy, also a former employee at the MPIIB. The researchers were able to show in a mouse model that a vitamin A deficiency, for example, leads to such a signal shift. As a result, squamous epithelial cells were activated in the inner cervix, and replaced the columnar epithelial cells which are normally present in this area.
"Our findings help to understand the mechanisms of the pathogenesis of cancer in these transition zones more precisely. They can also help to improve the early detection of cervical cancer, and to develop precise strategies for prevention and improved treatments," said Meyer. "As the next step, we are now trying to better understand the external influences and processes which lead to a signal change in the immediate cell environment, and thus to metaplasia formation. Preliminary findings also suggest a possible role of inflammation here. We'll investigate this in future studies in Kiel, also together with our colleagues at the Cluster of Excellence PMI," added Meyer.
Scientific contact:
Prof. Dr. Thomas F. Meyer
Infection Oncology working group
Institute of Clinical Molecular Biology (IKMB)
Kiel University and the University Medical Center Schleswig-Holstein
t.meyer@ikmb.uni-kiel.de
Illustration of the tissue in the cervix with squamous epithelia (green) of the outer cervix (ectocervical) and columnar epithelia (red) of the inner cervix (endocervical) merging at the transition zone. The opposing Wnt signaling environment maintains this distribution. A change toward signals in which Wnt signaling is suppressed in the endocervical stroma promotes the development of squamous metaplasia.
Original publication:
C. Chumduri*, R. K. Gurumurthy*…T. F. Meyer: Opposing Wnt signals regulate cervical squamocolumnar homeostasis and emergence of metaplasia. Nature Cell Biology (2021). https://doi.org/10.1038/s41556-020-00619-0
*These authors contributed equally to the work.
About the Cluster of Excellence PMI
The Cluster of Excellence "Precision Medicine in Chronic Inflammation" (PMI) is being funded from 2019 to 2025 through the German Excellence Strategy (ExStra). It succeeds the "Inflammation at Interfaces” Cluster, which was already funded in two periods of the Excellence Initiative (2007-2018). Around 300 members from eight institutions at four locations are involved: Kiel (Kiel University, University Medical Center Schleswig-Holstein (UKSH), Muthesius University of Fine Arts and Design, Kiel Institute for the World Economy (IfW), Leibniz Institute for Science and Mathematics Education (IPN)), Lübeck (University of Lübeck, University Medical Center Schleswig-Holstein (UKSH)), Plön (Max Planck Institute for Evolutionary Biology) and Borstel (Research Center Borstel - Leibniz Lung Center).
The goal is to translate interdisciplinary research findings on chronic inflammatory diseases of barrier organs to healthcare more intensively, as well as to fulfil previously unsatisfied needs of the patients. Three points are important in the context of successful treatment, and are therefore at the heart of PMI research: the early detection of chronic inflammatory diseases, the prediction of disease progression and complications, and the prediction of individual responses to treatment.
Press office
fbuhse@uv.uni-kiel.de+49 (0)431/880 4682 https://precisionmedicine.de
Cluster of Excellence "Precision Medicine in Chronic Inflammation"
Scientific Office
Head: Dr. habil. Susanne Holstein Postal
Christian-Albrechts-Platz 4, 24118 Kiel, Germany
Contact: Sonja Petermann
+49 (0)431 880-4850, fax: +49 (0)431 880-4894
spetermann@uv.uni-kiel.de
Twitter: PMI @medinflame